Generate clinically relevant quantities of stem cells with the beadMATRIX family.
beadMATRIX – developed for expansion of MSC.
beadMATRIX+ optimized for the culture of iPSC.
YOUR CHALLENGE
Your aim is to generate clinically relevant quantities of stem cells such as mesenchymal stromal cells (MSCs) or induced pluripotent stem cells (iPSC) with an expected application in cell-based therapies. You have faced difficulties upscaling your expansion process from 2D to 3D while maintaining cell quality, safety, and potency. Ideally, you would like to boost the efficiency of your manufacturing process while standardizing protocols and complying with safety regulations.
OUR SOLUTION
We are offering animal component-free, pre-coated microcarriers specifically designed for bioreactor-scale expansion of stem cells in serum-free and xeno-free media. The coating formulations are based on the 2D coating products of the myMATRIX family. While beadMATRIX is qualified to expand MSCs, beadMATRIX+ has been refined to optimally support iPSCs. These products are tailored to support customers in the establishment of translational cell therapies. Documentation describing quality and origin characteristics are provided.
YOUR BENEFITS
Large-scale production of stem cells with consistency across scales – benefit from our isoMATRIX, myMATRIX and beadMATRIX product ecosystem.
beadMATRIX
Enabling high yield and quality of MSCs
denovoMATRIX has developed a specific coating composition and performed an elaborated optimization process to ensure a high-performing microcarrier for the 3D culture of MSCs. Application of beadMATRIX accelerates the proliferation of highly viable and therapeutically active MSCs up to 25% compared to the leading brand competitor. Harvested cells show MSC-specific characteristics as defined by the International Society of Cellular Therapy such as distinctive CD-marker expression and trilineage differentiation potential. In addition, they present immunomodulatory potential and angiogenic activity.
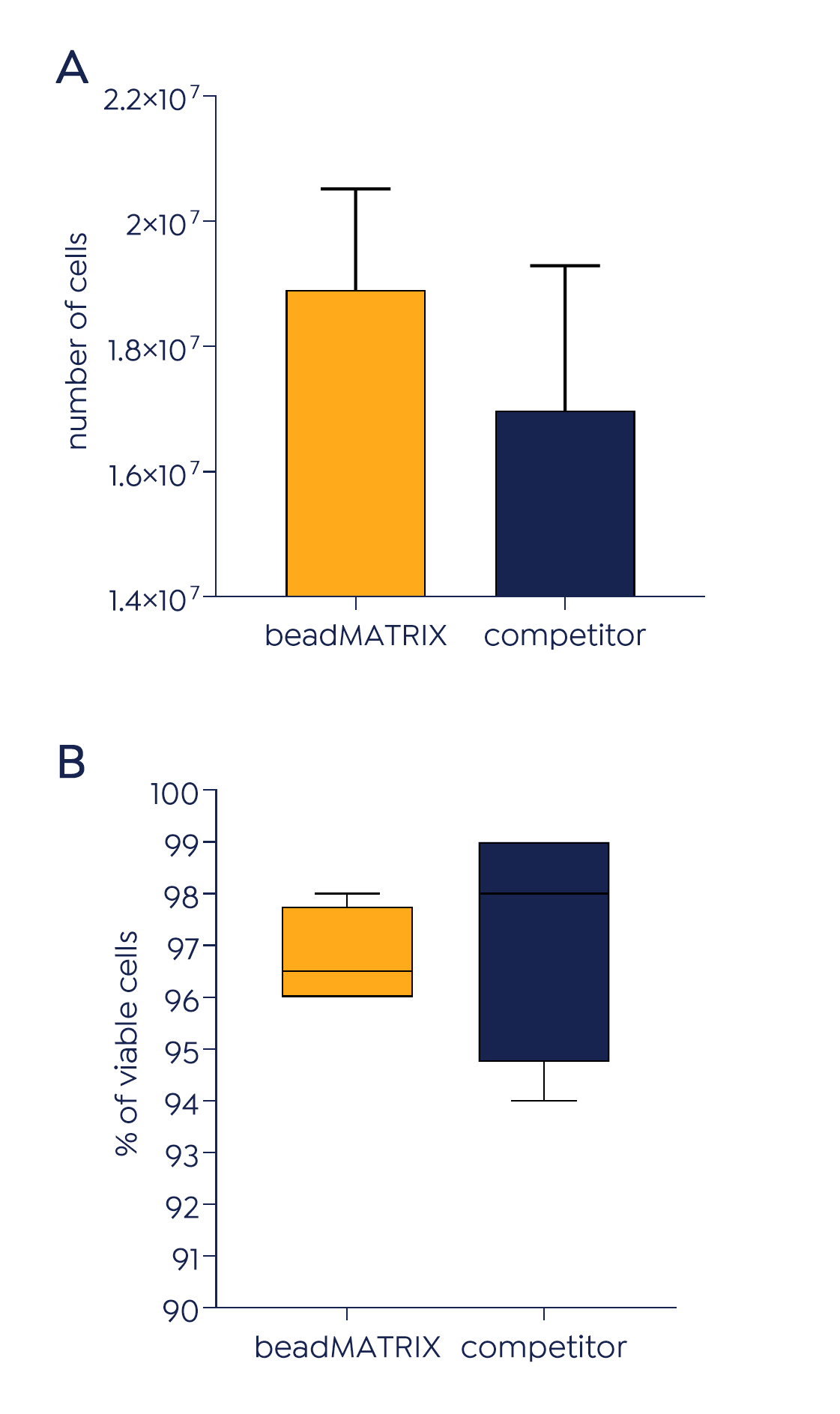
beadMATRIX+
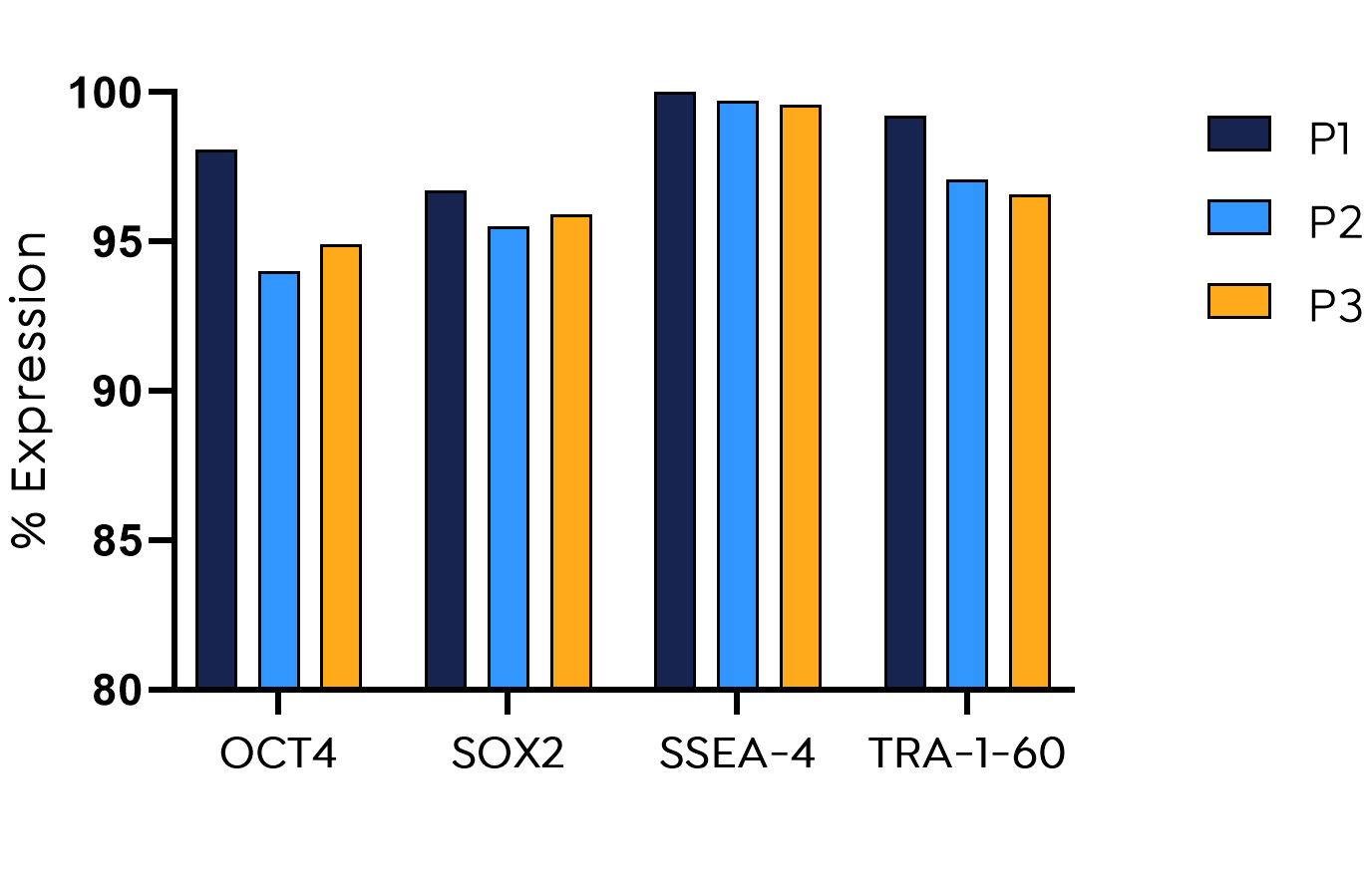
Supporting efficient 3D-culture of iPSC
The composition of the beadMATRIX coating has has been further improved to optimally support the expansion of iPSC. iPSCs cultured with beadMATRIX+ proliferate fast and reliable over 3 passages (21 days) while remaining pluripotent and genetically stable. High expression of the stemness genes OCT4, SOX2, SSEA-4, and TRA-1-60 is ensured over multiple passages in a 3D culture setup. The results show that beadMATRIX+ supports dynamic culture of iPSCs and can be implemented for efficient iPSC manufacturing for clinical applications.
Compatible with various manufacturing processes
The upscaling of cell manufacturing is a complex topic. We aim to support the establishment and improvement of existing processes with the help of a highly compatible product. Our beadMATRIX products have been successfully tested with different bioreactor setups and several cell culture media. Our production procedures ensure batch-to-batch consistency, product stability and compliance with the high-quality standards of ISO9001 and ISO14644. beadMATRIX and beadMATRIX+ documentation addresses pharma grade requirements in respect of material origin and quality. The microcarriers are sterile and ready-to-use and available in 10 gram units with larger sizes available upon request.
LET US DO THE HEAVY LIFTING.
We develop, characterize and manufacture extracellular matrix-mimetic coatings for your applications. denovoMATRIX also offers its expertise in cell line establishment, cell expansion and cell analysis to enhance your research and development projects. Contact us for more information on our service and licensing offer.

